Indigenous Australians have unique microbes in their mouths, which may contribute to a higher burden of chronic diseases, such as diabetes and cardiovascular disease, according to new research led by Penn State and the University of Adelaide. The results suggest that understanding this “oral microbiome” could be a useful target to help Indigenous Australians improve their health.
Like the gut microbiome is the community of microbes living in the gut, the oral microbiome is the community of microbes living in the mouth. Some of these oral microbes are crucial to human health; for example, they can defend against invading pathogens. Others, however, may be harmful and contribute to oral diseases, such as periodontal—or gum—disease.
“Indigenous Australians disproportionately experience poor oral health, which is linked to other negative health outcomes,” said Laura Weyrich, associate professor of anthropology, Penn State. “For example, periodontal disease increases the risk and severity of diabetes, chronic kidney disease and cardiovascular disease. Exploring oral microbiome may reveal new pathways to improve both oral and systemic health outcomes for Indigenous Australians.”
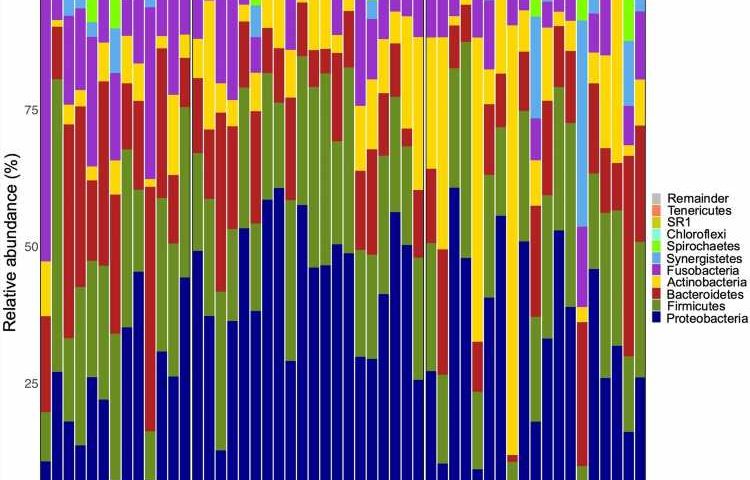
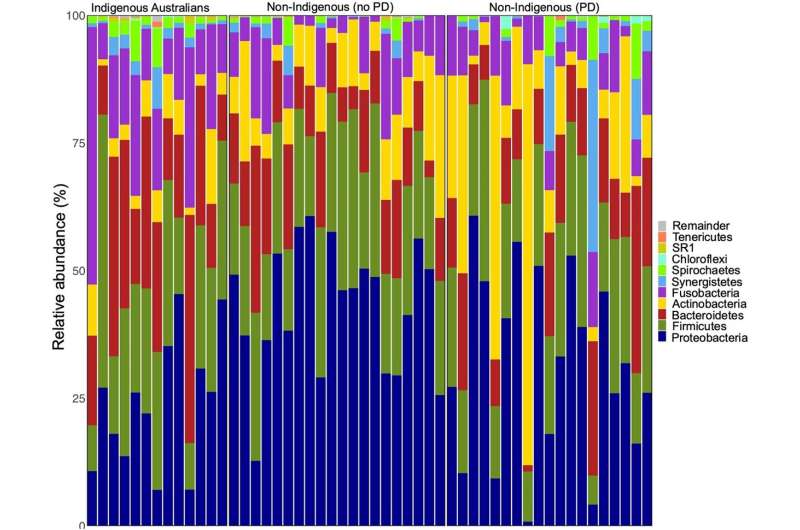
The researchers obtained samples of dental calculus—a form of hardened dental plaque—from 13 Indigenous Australians with periodontal disease, 19 non-Indigenous individuals with periodontal disease and 20 non-Indigenous individuals without periodontal disease. The Aboriginal Australian study participants hailed from multiple cultural/linguistic groups, with approximately half living in the arid Central Desert region of the Northern Territory and half in the tropical Top End—regions separated by approximately 930 miles.
They used a technique called 16S ribosomal RNA gene amplicon sequencing to characterize the types—or taxa—of microbes found in dental calculus. The findings were published in the journal Evolution, Medicine, and Public Health.
The team found that the composition of oral microbes differed significantly between the Indigenous and non-Indigenous individuals, with samples from Indigenous Australians containing several taxa—including Mogibacteriaceae, Porphyromonas, Tissierellaceae, Desulfomicrobiaceae and Methanobrevibacter—that may be associated with periodontal disease. For example, Porphyromonas gingivalis is thought to play an important role in tissue destruction in periodontal disease.
In addition, the researchers found that the oral microbiome samples from Indigenous Australians contained a greater diversity of microbial taxa than the samples from non-Indigenous individuals, including a relatively high proportion of microorganisms unique to that group. These included species of Endomicrobia, which are bacteria that live in the guts of termites and allow the insects to digest cellulose.
“Termites are a traditional food for some Aboriginal Australians in the Central Desert region and the Northern Territory,” said Weyrich. She explained that while bacteria can become associated with dental calculus as a result of a person’s current diet, some bacteria may be “inherited” through generations or as a result of their ancestors’ diets.
“The fact that we found termite-associated species in the oral microbiome of Aboriginal Australians who are not regularly engaging with these traditional foods or medicines suggests that heritage may play a role in the maintenance of Endomicrobia and other unique oral species,” she said. “This kind of “vertical transmission” may also be the source of some bacteria that may contribute to periodontal disease.”
The team also found that geography played a role in the taxa observed within the oral microbiota of Aboriginal Australians. For example, the termite-associated bacteria were most prevalent in the oral microbiome of Indigenous Australians living in Central Australia.
“Indigenous Australians have close connections to their geographic homelands, with many groups living in a particular location for 50,000 years or more,” said Weyrich. “During this time, Indigenous Australians’ microbiome could have adapted to specific environments or cultural practices such as diets or traditional medicines.”
“The Endomicrobia finding is quite an intriguing one,” said Matilda Handsley-Davis, who worked on the study as a doctoral candidate in biological sciences at the University of Adelaide. “It makes you think about all the stories the microbiome can hold about our relationships with our family, our ancestors and the environment around us. It would be great to see this idea explored further through community-based research partnerships in the future, perhaps incorporating community knowledge about, for example, dietary practices and connection to the landscape via the microbiome.”
Source: Read Full Article