A local study led by the Agency for Science, Technology and Research (A*STAR) discovered that by inhibiting the function of two proteins, G9a and GLP, during the cell therapy production process, immune cells could become more effective in combatting cancer. These findings, published in the journal Nature Communications, can help advance the development of innovative therapies that could benefit cancer patients, bringing us closer to more effective targeted treatments for solid tumor cancers.
Solid tumors are a major cause of cancer-related deaths worldwide. Traditional treatments such as chemotherapy, radiation therapy and surgery are available, but they have differing efficacy against solid tumors, particularly in advanced stages of the cancer. T-cell therapy has been very successful in targeting liquid tumors such as blood cancers, but the same efficiency has not been observed in solid tumors such as breast, liver or brain cancer.
Engineered T cells are usually introduced into the patient’s bloodstream as part of the treatment. They are in the same environment as liquid tumors, allowing them to locate and target the liquid tumors easily. However, in the case of solid tumors, the engineered T cells face physical and molecular obstacles such as migrating through dense tissue structure inside the body and encountering other cells and molecules that may negatively impact their function.
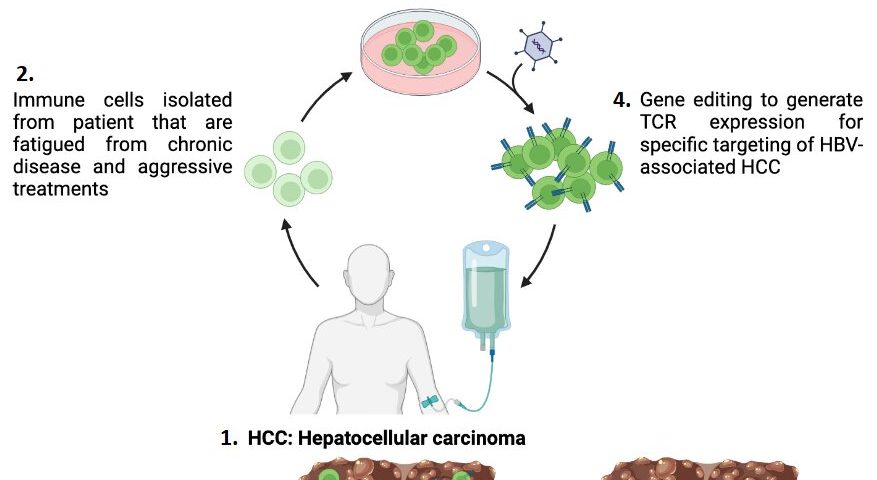
Researchers and clinicians from A*STAR’s Institute of Molecular and Cell Biology (IMCB) and Singapore Immunology Network (SIgN), and Duke-NUS Medical School collaborated to explore innovative approaches to improve the efficiency of T cells, the immune cells responsible for recognizing and eliminating cancer cells.
The research team, led by Dr. Andrea Pavesi, Senior Scientist at A*STAR’s IMCB, conducted a comprehensive analysis of epigenetic drugs that can affect the efficacy of the engineered T cells in increasing anti-tumor activity. The team used 2D and novel 3D assays that mimicked the physical environment that T cells would encounter to find and target the cancer cells in the human body.
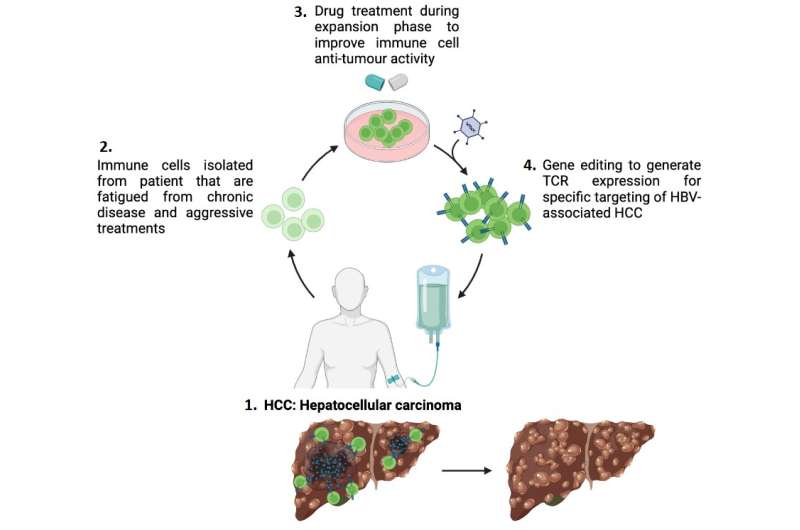
A drug was administered to the immune cells during the cell expansion process of cell therapy performed in the lab, which targeted the G9a and GLP proteins. The drug was subsequently washed away before the engineered immune cells were re-introduced into the patient’s body, thus eliminating side-effects from the drug. The findings showed that the drug helped to increase the anti-tumor function of the engineered immune cells—it increased the production of granzymes, proteins that help to locate and eliminate target tumor cells.
Tapping on the immune cell profiling capabilities of Dr. Giulia Adriani, Principal Scientist at A*STAR’s SIgN, and patient samples from Duke-NUS, the study’s findings were validated using well-established cell-lines and patient-derived immune cells to confirm the efficacy of blocking G9a and GLP activity in improving the efficiency of T-cell therapy. The results showed that the drug enhanced the anti-tumor function of engineered immune cells.
This would mean better patient outcomes such as improved survival rates and quality of life. It also has broad implications for all cell therapies targeting solid tumors. Patients with a weak immune system, who usually require immune cells from healthy donors for cell therapy treatment, may also benefit from treatments using their own immune cells.
This reduces the chances of the patient’s body rejecting the cells, as well as implications from using incompatible cells. The drug used to block G9a and GLP activity also holds significant potential for further development, presenting itself as an attractive therapeutic option for cancer treatments.
Dr. Andrea Pavesi, Senior Scientist at A*STAR’s IMCB and lead author of the study, said, “The approach of improving the individual anti-tumor activity of each immune cell can address many limitations in T-cell therapy and enhance treatment efficacy. Our discovery will advance the development of effective therapeutics for solid tumor cancers and help improve lives.”
Professor Antonio Bertoletti, from Duke-NUS’ Emerging Infectious Diseases Programme, said, “There is a high demand for the production of suitable T cells for adoptive T-cell therapy, a type of cell therapy where engineered T cells are administered to patients to fight diseases such as cancer. This discovery could help improve cell therapies that use both patient-derived and donor-derived immune cells, benefitting a variety of patients. We hope to work towards successful clinical trials and bring this method to market to improve patient outcomes.”
More information:
Maxine S. Y. Lam et al, G9a/GLP inhibition during ex vivo lymphocyte expansion increases in vivo cytotoxicity of engineered T cells against hepatocellular carcinoma, Nature Communications (2023). DOI: 10.1038/s41467-023-36160-5
Journal information:
Nature Communications
Source: Read Full Article